COVID Testing Kits
- (1)
- (9)
- (1)
- (1)
- (1)
- (2)
- (5)
- (1)
- (1)
- (2)
- (1)
- (2)
- (2)
- (3)
- (36)
- (1)
- (1)
- (5)
- (1)
- (1)
- (1)
- (1)
- (6)
- (1)
- (5)
- (2)
- (17)
- (3)
- (5)
- (2)
- (5)
- (2)
- (1)
- (7)
- (22)
- (13)
- (2)
- (6)
- (1)
- (5)
- (3)
- (10)
- (8)
- (1)
- (5)
- (1)
- (2)
- (4)
- (9)
- (1)
Filtered Search Results
Cayman Chemical SARS-CoV-2 nutralIzng ntIb 1ea
SARS-CoV-2 Neutralizing Antibody (B.1.351) Positive Control has been tested and formulated to work exclusively with Cayman’s SARS-CoV-2 (B.1.351, Beta Variant) Neutralizing Antibody SimpleDetect ELISA Kit (Item No. 502200).

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Sino Biological SARS-CoV-2 Spike S1+S2 (D614G) trimer Protein (ECD, His tag) (HPLC-verified), Endotoxin-Free
A DNA sequence encoding the SARS-CoV-2 Spike S1+S2 (YP_009724390.1, with mutations D614G, F817P, A892P, A899P, A942P, K986P, V987P and furin cleavage site mutants) expressed with the bacteriophage T4 fibritin and a polyhistidine tag at the C-terminus.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
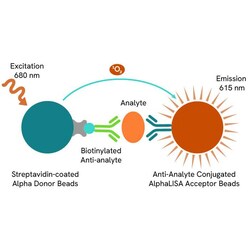
Revvity Health Sciences Inc AlphaLISA SARS-CoV2 Nucleocapsid Detection Kit, 5,000 Assay Points
AlphaLISA SARS-CoV2 Nucleocapsid Detection Kit, 5,000 Assay Points

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
IHEALTH LABS INC iHealth Flu A&B/COVID-19 3-in-1 Rapid Test Pro (25 Pack) - Point of Care
The iHealth COVID-19/Flu A&B Rapid Test Pro is a lateralflow immunochromatographic assay intended for in vitro rapid, simultaneous qualitative detection and differentiation of influenza A and influenza B nucleoprotein antigens and SARS-CoV-2 nucleocapsid antigen directly from anterior nasal swab specimens of individuals with signs and symptoms of respiratory infection consistent with COVID-19 by their healthcare provider within the first four (4) days of symptom onset when tested at least twice over three days with at least 48 hours between tests. Clinical signs and symptoms of respiratory viral infection due to SARS-CoV-2 and influenza can be similar. Testing is limited to laboratories certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA), 42 U.S.C. § 263a, that meet the requirements to perform moderate, high or waived complexity tests.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Meso Scale Discovery R-PLEX SARS-CoV-2 Spike ASYQP
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
The R-PLEX SARS-CoV-2 Spike Assay is a multiplex immunoassay panel that detects antibodies to the SARS-CoV-2 spike protein in human serum and plasma It uses Meso Scale Discovery electrochemiluminescence technology and provides quantitative results for IgG IgM and IgA antibodies The assay has high sensitivity and specificity and is intended for use as an aid in identifying individuals with an adaptive immune response to SARS-CoV-2 indicating recent or prior infection It requires a small sample volume and provides results in under 3 hours The kit includes a 96-well plate calibrators controls diluent and substrate

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Sino Biological SARS-CoV-2 (2019-nCoV) Spike S1+S2 ECD (R683A, R685A, F817P, A892P, A899P, A942P, K986P, V987P)-His Recombinant Protein (HPLC-verified), Endotoxin-Free
A DNA sequence encoding the SARS-CoV-2 (2019-nCoV) Spike Protein (S1+S2 ECD) (YP_009724390.1) (Met1-Pro1213(R683A, R685A, F817P, A892P, A899P, A942P, K986P, V987P)) was expressed with the bacteriophage T4 fibritin and a polyhistidine tag at the C-terminus.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
AdipoGen SARS-CoV-2 Neutralizing Antibodies Detection Kit (96 wells)
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
SARS-CoV-2 contains 4 structural proteins, including Envelope (E), Membrane (M), Nucleocapsid (N) and Spike (S), which is a transmembrane protein, composed of two subunits S1 and S2. The S1 subunit contains a receptor binding domain (RBD), which binds to the cell surface receptor Angiotensin-Converting Enzyme 2 (ACE2) present at the surface of epithelial cells, causing mainly infection of human respiratory cells.The SARS-CoV-2 Neutralizing Antibodies Detection Kit contains key reagents required to test the presence of functional neutralizing antibodies against SARS-CoV-2 present in the serum or plasma. It is an easy and fast alternative to the classical neutralization assay using Vero E6 cells. This Detection Kit is based on a colorimetric reaction, which measures the binding of the RBD of the Spike S protein from SARS-CoV-2 to its human receptor ACE2.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Bioss SARS-CoV-2 2019-nCoV Nucleocap
SARS-CoV-2 2019-nCoV Nucleocapsid Monoclonal Antibody

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Sigma Aldrich Fine Chemicals Biosciences Prostate Specific Antigen
Prostate specific antigen (PSA) is a 28.43 kDa single-chain glycoprotein containing 237 amino acids encoded by the kallikrein gene mapped to human chromosome 19. It is specifically expressed by human prostate tissue and is one of the highly abundant proteolytic enzyme expressed in the seminal fluid.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Bioss SARS-CoV-2 S-Protein ACE2 Bind
SARS-CoV-2 S-Protein ACE2 Binding Domain Primary Antibody Unconjugated Host Species Mouse Target Species SARS-CoV-2 Clonality Monoclonal Applications WB 2G1Concentration Lot Dependent Size 100 ul Storage Buffer PBS 50% glycerol and 5 mm NaN3

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Genscript Corporation SARS-CoV-2 Nucleocapsid Antibo
The product is specific for SARS-CoV-2 Nucleocapsid protein

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Medchemexpress LLC SARS-CoV-2-IN-6 | 2725749-22-2 | 99.9% | 312.75 g·mol-1 | C17H13ClN2O2 | 5 MG
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
SARS-CoV-2-IN-6 is a research-use small-molecule inhibitor targeting the SARS-CoV-2 3C-like protease (3CLpro). It inhibits protease enzymatic activity with a reported IC50 of 73 nM, is supplied as a small milligram pack for laboratory studies, and is accompanied by standard product documentation and safety information.
- Potent enzymatic inhibition with IC50 of 73 nM.
- Designed to target the SARS-CoV-2 3C-like protease (main protease).
- Molecular formula C17H13ClN2O2 and molecular weight 312.75 g·mol-1.
- High purity suitable for research applications (≈99.9% by HPLC).
- Provided in small research-scale pack sizes for assay development.
- Intended for laboratory research use only; not for human therapeutic use.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
IVANO BIOSCIENCE Validated SARS-CoV-2 pseudovirus, Beta B.1.351 variant, 100 µL of pseudovirus enables 50 reactions in a 96-well plate.
Recombinant 3rd-generation lentiviral vector displaying the spike protein (S) of the SARS-CoV-2, Beta B.1.351 variant. It incorporates a luciferase reporter gene.Performance: 2 µL per well (96-well plate) produces a signal-to-background ratio of 10³ RLU, according to the manufacturer’s protocol. 100 µL = 50 reactions.Validation: Validated lot-to-lot for infection and neutralization.Applications: This pseudovirus can be used to investigate virus entry, receptor recognition, and cellular tropism, to perform functional studies, to screen anti-S drugs and neutralizing antibodies, or to assess vaccine efficacy. For Research Use Only.Biosafety: Must be handled under BSL-2 conditions. Replication-incompetent and non-pathogenic due to multiple molecular safety modifications.Shipping and storage: Shipped on dry ice and stored at −80 °C. Avoid freeze/thaw cycles. Each shipment includes a complete protocol and a batch-specific quality control report.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Medchemexpress LLC SARS-CoV-2 spike receptor-binding domain (Omicron, B.1.1.529) | >95.0% | 10 UG
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Recombinant SARS-CoV-2 spike receptor-binding domain (Omicron B.1.1.529), expressed in mammalian cells and purified for use in biochemical and immunological assays. The protein carries a C-terminal His tag, is supplied as a sterile, filtered PBS solution, and is intended for applications such as ELISA, binding studies, and antibody characterization.
- Expressed in HEK293 cells with a C-terminal His tag.
- Represents the Omicron (B.1.1.529) variant of the spike RBD.
- Purity greater than 95% by reducing SDS-PAGE.
- Supplied as a 0.22 μm filtered PBS solution, pH 7.4.
- Store at -80°C; shipped on dry ice; avoid repeated freeze-thaw cycles.
- Suitable for ELISA, binding assays, and antibody characterization.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
ATILA BIOSYSTEMS INC iAMP COVID-19 Detection Kit
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
The iAMP COVID-19 Detection Kit is a real-time reverse transcription isothermal amplification test. The test is based on a proprietary isothermal amplification technology termed OMEGA amplification (Patent: WO 2017/205510 A1; publication: The Journal of Molecular Diagnostics, Vol.22, No 3, 419-428, 2020). OMEGA primer sets are designed to specifically detect RNA and later cDNA from the N and ORF-1ab genes of the SARS-CoV-2 virus in nasal, nasopharyngeal and/or oropharyngeal swabs from patients with signs and symptoms of infection who are suspected of COVID-19. The iAMP COVID-19 assay’s key differentiator from current rRT-PCR COVID-19 assays is its ability to detect SARS-CoV-2 RNA directly from samples without prior RNA extraction process. Swab specimens are inserted directly into our 1X iAMP COVID-19 Sample Buffer Mix with a 15 min incubation at room temperature and can be directly used for OMEGA isothermal amplification and signal detection. Sample to result takes about 1 hour.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More